How is Ammonium Nitrate Produced Industrially?Īmmonium nitrate is produced industrially by the reaction of ammonia and nitric acid. Where Does Ammonium Nitrate Occur Naturally?Īmmonium nitrate is used as a fertilizer and it can also be found in some explosives. The molecular weight of ammonium nitrate is 90.03 grams/mole. 
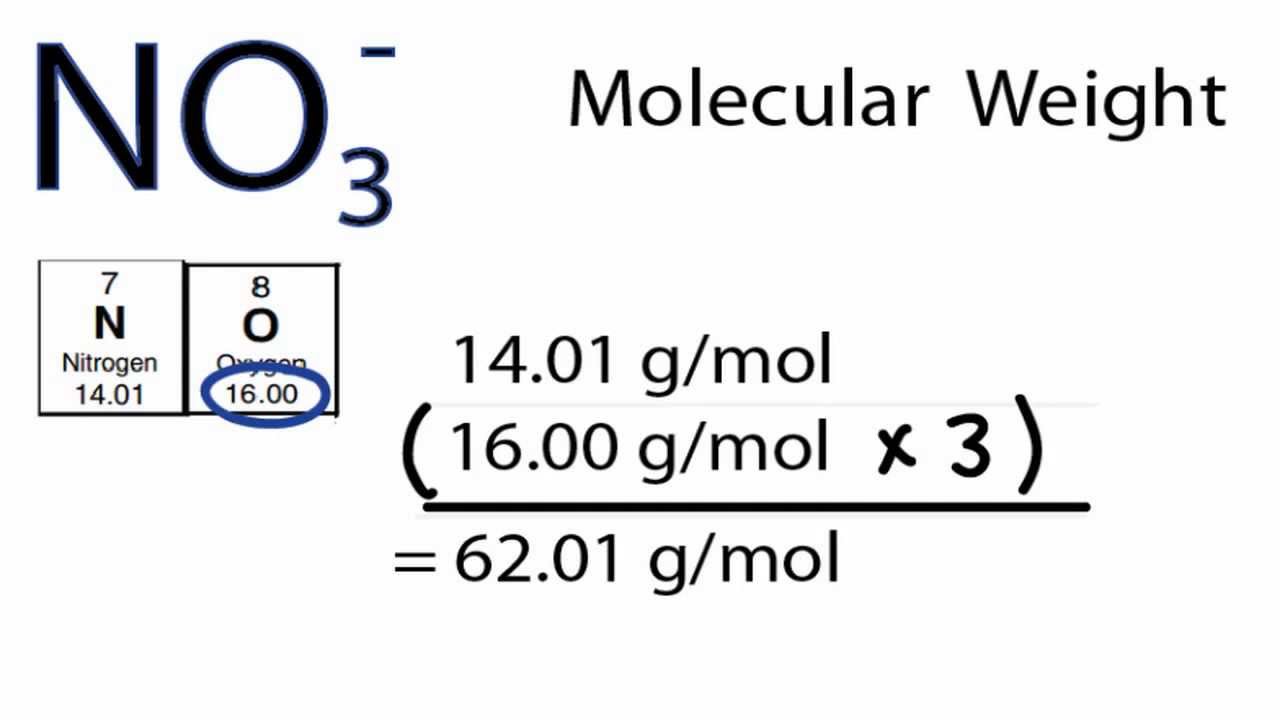
The molecular weight of ammonium nitrate can be calculated by adding the atomic weights of nitrogen (14.01) and oxygen (16.00) and then multiplying the sum by the number of atoms in the molecule (3). How to Calculate the Molecular Weight of the Ammonium Nitrate? Ammonium Nitrate Element Mass Percent Oxygen 47.9982g Oxygen 47.9982g Nitrogen 28.0134g Nitrogen 28.0134g Hydrogen 4.0318g Hydrogen 4. Next, using the periodic table, find the atomic mass in g/mol of each element (the molar mass of an element is equal to its atomic mass. The molar mass of ammonium nitrate is 80 grams/mol. The first step to finding the molar mass of Ceric Ammonium Nitrate is to count the number of each atom present in a single molecule. What is the Molar Mass of Ammonium Nitrate? The molar mass and molecular weight of N 2 H 4 O 3 is 80.043. The physical properties of ammonium nitrate are that is is a white crystalline solid, it is soluble in water, it is a strong oxidizer, and it is explosive in the presence of heat or a spark. 
What are the Physical Properties of Ammonium Nitrate? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
